Corrosion Presents in Different Types of Rust
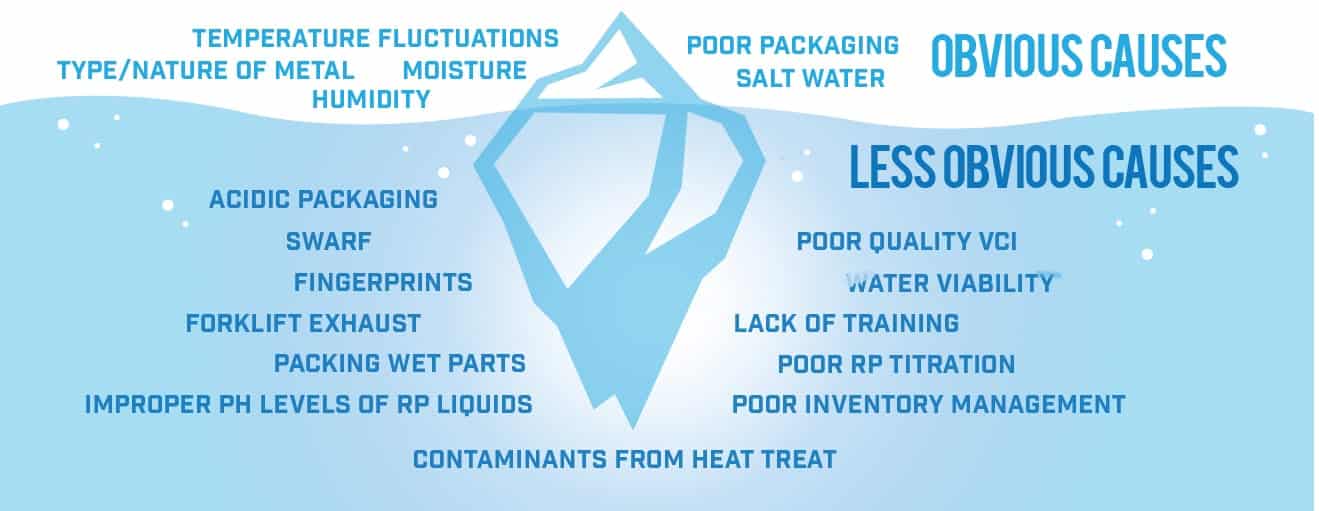
They are different in how they look and different in the circumstances that led to their formation. At Armor Protective Packaging, we recognize that identifying the type of rust is the first step in identifying a solution to prevent it. We like to compare rust to an iceberg (see Rust Iceberg graphic below) — it’s not what you can see above the surface of the water that is most destructive, but rather it’s what you can’t see that can do the most harm. The same is true of rust and corrosion, often it’s the things that aren’t visible or that are less obvious that result in the greatest damage.
Visually, rust appears in a color spectrum that can range from yellow to red to black. Conditions that lead to rust formation also run the spectrum, but most can be attributed to one of three general factors:
Process Related
Whether it is the cleaning process, the metal fabrication process such as cold working, heat treating or machining or the handling process, this aspect of producing a metal part is a primary contributor of corrosion. Much of what is seen in terms of corrosion problems, happen inside the manufacturing plant before it’s even packaged. With so many variables inside of a manufacturing setting, maintaining tight control of your processes is critical to preventing rust.
Packaging Related
The type of materials used to package metal parts is important. Acidic materials such as corrugated and non-treated paper can actually trap or hold moisture and cause corrosion. Sometimes in a companies’ desire to fix a corrosion problem they add more packaging, which can have a reverse effect by contributing even more to the rust problem.
Environmental
While the weather (temperature and humidity) can certainly be the most obvious environmental factor, other things such as contaminants in the air pose equal areas of concern.
Identifying the type of rust you are experiencing can provide valuable insight as to what may be its root cause. To help, ARMOR developed a roster of the types of rust that are frequently found in the manufacturing or outbound shipping setting — complete with photos and the key attributes and contributors of each type of rust.

Red Rust
Hydrated oxide Fe2O3•H2O (high oxygen/water exposure)
Rust from Iron (III) oxides forms due to high oxygen and water exposure resulting in red rust.
- Red rust is the result of heavy exposure to air and moisture, combined many times with a contaminate (salt).
- This type of rust is most likely atmospheric because typically there are no signs of rust runs or streaks on the metal parts/equipment where the rust has formed.
- With red rust, there is uniform corrosion, most often from a very corrosive environment.

Yellow Rust
Iron oxide-hydroxide FeO(OH)H2O (high moisture)
Rust from Iron (III) oxides that is a very soluble iron oxide results in yellow rust.
- Yellow rust is distinguishable in recessed areas of the metal parts/equipment where the rust “runs and drips” (solvated rust).
- Yellow rust forms as a result of very high moisture content. It frequently found in settings where puddled/standing water has most likely been present.

Brown Rust
Oxide Fe2O3 (high oxygen/low moisture)
Rust from Iron (III) oxides with high oxygen and low moisture results in brown rust.
- Brown rust is a drier rust than those mentioned above.
- It is most likely atmospheric – having formed as a result of water and oxygen in the atmosphere and presenting as a reddish-brown crust on the metal’s surface.
- Brown rust is sometimes localized rust which appears as non-uniform spots or only in certain areas rather than over the whole surface. It can be the result of a contaminate on the metal’s surface often originating from the manufacturing process.

Black Rust
Iron (II)oxide – Fe3O4 (limited oxygen)
Rust from Iron (III) oxides with limited oxygen and low moisture results in black rust.
- Black rust can be visually identified as a thin, black film which is the result of oxidation in a low oxygen environment.
- Black rust has an appearance of almost a black stain. Most likely the areas exhibiting the black rust had something covering them, which prevented oxygen from reaching the surface.
- This type of rust is a more stable rust layer that does not propagate as rapidly as other rust forms.

Multiple Forms
Multiple forms of corrosion can be present at once
- Brown rust has a lower moisture content and is most likely the result of atmospheric conditions.
- Settings where black rust might form include wet paper resting tightly against a metal surface.
- Yellow rust has a high moisture content and can frequently be attributed to an environment where water is most likely present.
EXPERIENCING RUST PROBLEMS?
Contact an ARMOR corrosion specialist to schedule a visit for a corrosion audit.





